
The ČSN EN ISO 13485:2016 standard was prepared in view of the higher demands placed on the quality management systems of the suppliers and manufacturers of medical devices. The EN ISO 13485:2003 standard was published as a harmonized standard for the European Directives 93/42/EEC and 90/385/EEC in the Official Journal of the European Union, which enables its use to demonstrate conformity with the requirements of these European Directives. We offer you the opportunity to take advantage of our experience, knowledge and technical equipment as well 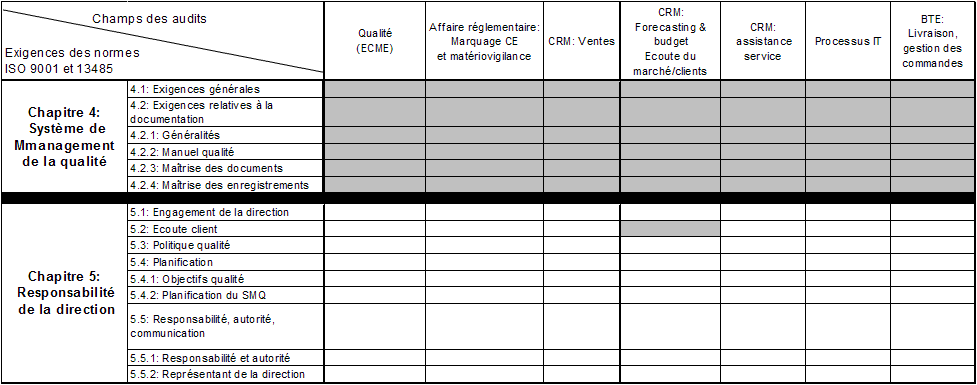
The Electrotechnical Testing Institute has a long tradition and experience in the certification of medical devices and verification of the quality management systems of their manufacturers and suppliers.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
